IMAGE. Self Check Image 1.
|
|
|
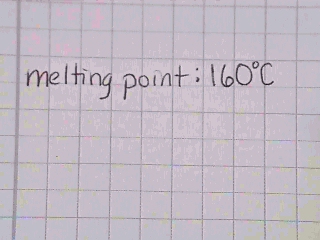
IMAGE. Self Check Image 2.
|
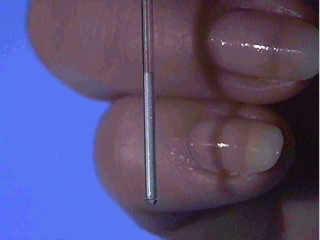
 Why is this sample in Self Check Image 1 not appropriate for use in a melting point experiment?
Answer
Why is this sample in Self Check Image 1 not appropriate for use in a melting point experiment?
Answer
The capillary tube contains too much sample for use in the Mel-Temp™. The tube should only have about 1-2 mm of sample in it.  You carefully observe the melting range of a sample of salicylic acid that you
prepared in the lab to be 140.7 to 149.3 °C. The known value of the melting point is 159
°C. What does the melting range you observed tell you about your sample?
Answer
You carefully observe the melting range of a sample of salicylic acid that you
prepared in the lab to be 140.7 to 149.3 °C. The known value of the melting point is 159
°C. What does the melting range you observed tell you about your sample?
Answer
Since the melting range is broad and much lower than the expected value for the melting point, the sample is probably quite impure. 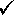 What melting range should you record for the sample shown in Self Check Video 1?
Answer
What melting range should you record for the sample shown in Self Check Video 1?
Answer
The sample first begins to turn to a liquid at 61.3 °C, and at about 64.2 °C the sample has completely melted. The melting range should be reported as 61.3 - 64.2 °C. 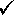 What might cause you to be concerned about the validity of the data page shown
in Self Check Image 2?
Answer
What might cause you to be concerned about the validity of the data page shown
in Self Check Image 2?
Answer
There is not enough information recorded. The data that is recorded might indiate one or more of the following:
Since the last digit recorded is supposed to be recorded by estimating between the graduation marks, recording 160 °C implies that the thermometer was graduated every 10 degrees. Commonly thermometers in the laboratory are more accurate than this.
The melting point was very sharp. Although if a sharp melting point was observed this observation should be recorded in the notebook. The standard practice is to record a melting point range (the temperature where the sample appears wetted and the temperature when the sample is completely liquid).
Since the last digit recorded is supposed to be recorded by estimating between the graduation marks, the melting point recorded is very approximate. |